What are Silicones?
Silicones are polymers that include synthetic compounds made up of repeating units of two silicon atoms and one oxygen atom frequently combined to form a siloxane backbone (⋯-Si-O-Si-⋯) with organic and or /hydrogen side groups attached to the tetravalent silicon atoms. In some cases, organic side groups can be used to link two or more of these -Si-O- backbones together. By varying the -Si-O- chain lengths, side groups, and cross-linking, silicones can be synthesized with a wide variety of properties and compositions. They can vary in consistency from liquid to gel to rubber to hard solids. Their chemical inertness, resistance to water and oxidation, and stability at both high and low temperatures have led to a wide range of commercial applications, from lubricating greases to electrical-wire insulation. They are typically rubber -like and heat-resistant, and are used in sealants, adhesives, lubricants, medicine, cooking utensils, and thermal and electrical insulation. Some common forms include silicone oil, silicone grease, silicone rubber, silicone resin and silicone caulk.
![]()
Properties of Silicones
Silicones exhibit many useful characteristics, including:
- Thermal stability (constancy of properties over a wide temperature range of −100 to 250°C).
- Resistance to oxygen, ozone, and ultra violet(UV) light. This property has led to widespread use of silicones in the construction industry (e.g. coatings, fire protection, glazing seals) and the automotive industry (external gaskets, external trim).
- Electrical insulation properties. Because silicone can be formulated to be electrically insulating or conductive, it is suitable for a wide range of electrical applications.
- Low thermal conductivity.
- Low chemical reactivity.
- Low toxicity.
- The ability to repel water and form watertight seals.
- Does not stick to many substrates, but adheres very well to others, e.g. glass.
- Does not support microbiological growth.
High gas permeability: at room temperature (25 °C), the permeability of silicone rubber for such gases as oxygen is approximately 400 times that of butyl rubber, making silicone useful for medical applications in which increased aeration is desired. Consequently, silicone rubbers cannot be used where gas-tight seals are necessary.
Categories /Forms of Silicones
Silicones are divided in to many classes namely:
- Silicone Fluids
- Silicone Elastomers
- Silicone Resins
- Silicone Greases
- Silicone Coatings
Applications and Uses
Polysiloxanes are manufactured as fluids, elastomers or resins, depending on the molecular weight of the polymers and the degree to which the polymer chains are inter-linked. Non-vulcanized, low-molecular-weight polysiloxane fluids are exceptionally stable to decomposition by heat, water, or oxidizing agents and are good electrical insulators. They make excellent lubricants and hydraulic fluids, as well as emulsions for imparting water repellency to textiles, paper, and other materials.
Silicones elastomers are used in many products in following applications: Electrical (e.g., insulation, potting, encapsulation), electronics (e.g., protective/conformal coatings), household (e.g., sealants for cooking apparatus), automobile (e.g., gaskets), aeroplane (e.g., seals), office machines (e.g., keyboard pads), medicine/dentistry (e.g., dental impression molds), textiles/paper (e.g., coatings), mold making(Ex: Prototyping).
Silicone resins are used in protective coatings and electrically insulating varnishes and for laminating glass cloth.
Composition, Structure and Properties
The silicones differ from most industrial polymers in that the chains of linked atoms that make up the backbones of their molecules do not contain carbon, the characteristic element of organic compounds. This absence of carbon in the polymer backbone turns polysiloxanes into unusual “inorganic” polymers and the organic groups, usually vinyl (CH=CH2), methyl (CH3), or phenyl (C6H5), are attached to each silicon atom. A general formula for silicones is (R2SiO)x, where R can be any one of a variety of organic groups.
The most common silicone compound, poly-dimethylsiloxane, can illustrate the central characteristics of this class of materials. The starting material is metallic silicon, which is obtained from silica (sand). Silicon metal is reacted with methyl chloride (CH3Cl) in the presence of a copper catalyst, forming dimethyldichlorosilane ([CH3]2Si[Cl]2). By reacting this compound with water, the chlorine atoms are replaced by hydroxyl (OH) groups. The resultant unstable compound, silanol ([CH3]2Si[OH]2), polymerizes in a condensation reaction, the single-unit molecules linking together to form poly-dimethylsiloxane (PDMS).. The di-methylsiloxane repeating unit of the polymer has the following structure:
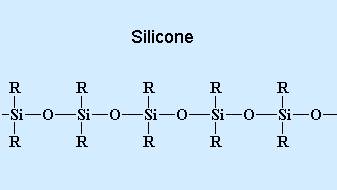
Siloxane molecules rotate freely around the Si-O bond, so, even with vinyl, methyl, or phenyl groups attached to the silicon atoms, the molecule is highly flexible. In addition, the Si-O bond is highly heat-resistant and is not readily attacked by oxygen or ozone. As a result, silicones are remarkably stable, and they have the lowest glass-transition temperature (the temperature below which the molecules are locked in a rigid, glassy state) and the highest permeability to gases of any polymer. On the other hand, the Si-O bond is susceptible to hydrolysis and attack by acids and bases, so silicones are relatively weak and readily swollen by hydrocarbon oils.